- Call / Whatsapp : +92 321 8467 101
- PKR - Pakistan
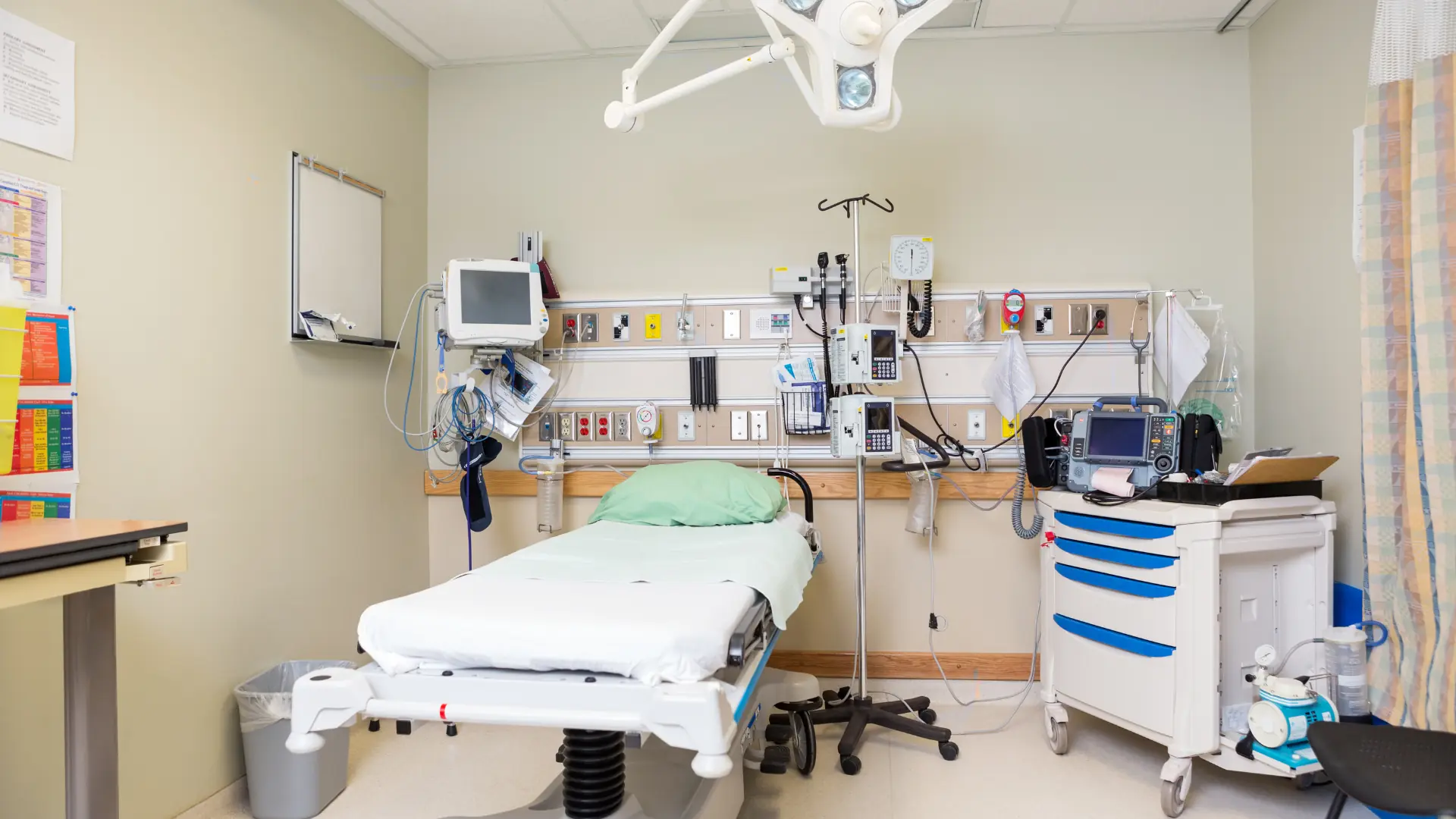
Medical video devices—cameras, monitors, and recording systems—drive contemporary medicine, from surgeries to diagnostics. But with patients' lives at stake, safety is not discretionary. These devices are subject to strict regulations, which guarantee they safeguard patients, clinicians, and data while providing consistent performance. It may seem overwhelming to wade through these regulations, but knowing them is essential to selecting equipment that works and meets the requirements. What are these safety standards, and why are they important? Let's dissect them, examining their intent, major requirements, and practical impact in healthcare.
Medical video equipment is not merely technology—it's a matter of life and death. A 2023 FDA report cited more than 500 adverse events attributed to defective medical devices, including imaging malfunctions. Safety standards exist to prevent such risks, covering electrical risk, radiation exposure, infection control, and data security. For hospitals, compliance equals avoiding fines and lawsuits, as well as preventing harm; for patients, it's the difference between a successful procedure and an avoidable complication. Whether it's a 4K endoscopy camera or a cleanroom monitor, these standards provide trust and precision. Here's what you need to know.
Core healthcare safety standards are a collection of essential guidelines and policies aimed at safeguarding patients, healthcare professionals, and confidential information during the entire care process. They include broad areas of protection such as patient data privacy (e.g., HIPAA adherence), infection prevention, medication safety, handling of equipment, and cybersecurity protocols. They promote healthcare practices to be consistent, evidence-based, and compliant with regulations. By following these safety standards, healthcare organizations can reduce risks, avoid medical errors, and ensure trust with patients and regulatory agencies. In the end, these standards are the foundation of a safe, dependable, and high-quality healthcare delivery system.
The International Electrotechnical Commission's IEC 60601-1 is the gold standard for medical electrical equipment. This critical standard ensures the safety, performance, and reliability of electronic devices used in clinical environments, including cleanroom monitors. Compliance with IEC 60601-1 means that equipment has been rigorously tested for electrical safety, mechanical integrity, and resistance to environmental factors. For hospitals and surgical centers, choosing monitors that meet this certification not only minimizes risk but also ensures alignment with global regulatory requirements, reinforcing a foundation of safe, high-quality patient care.
Purpose: Prevents shocks, burns, or fires due to faulty wiring.
Key Requirements: Grounding, leakage current limits (e.g., <0.5 mA for patient-contact devices), and insulation.
Healthcare Impact: Prevents a monitor in the OR from sparking or malfunctioning during surgery.
A defibrillator monitor to IEC 60601-1 standard can operate safely next to a patient, even when there's a power surge.
Keyword Tip: "IEC 60601-1 medical video" and "electrical safety in healthcare" are a natural fit here.
Video gear such as fluoroscopes discharges radiation, so requirements like ALARA (As Low As Reasonably Achievable) come into play. This safety principle mandates that radiation exposure to patients and staff is minimized without compromising diagnostic quality. When selecting equipment like monitors used alongside fluoroscopy, it’s essential they support precise imaging at lower radiation doses. Features like high-resolution displays, real-time processing, and compatibility with dose-reduction technologies help meet ALARA goals—enhancing safety while maintaining the clarity needed for accurate medical evaluations.Purpose: Lowers exposure to patients and personnel.
Key Requirements: Dose monitoring, automatic shutoffs, and shielding.
Healthcare Impact: Decreases cancer risks of repeated imaging.
A low-dose X-ray machine following FDA restrictions reduces exposure by 20%, according to a 2022 radiology study, without compromising image quality.
Keyword Tip: "Radiation safety medical video" and "ALARA healthcare standards" enhance relevance.
ISO 13485 is a globally accepted standard that provides the requirements for an overall quality management system specially designed for the design, development, manufacture, installation, and servicing of medical devices. ISO 13485 is important to ensure that medical devices have consistent quality and safety standards throughout their life cycle. It provides a structured framework for manufacturers to demonstrate their ability to produce devices that not only comply with regulatory requirements but also meet customer expectations and perform reliably in clinical environments.
Purpose: Ensures consistent safety and performance.
Key Requirements: Risk management, validation testing, and traceability.
Healthcare Impact: Guarantees a camera’s reliability across thousands of uses.
A MedImaging endoscopy system certified under ISO 13485 delivers dependable visuals every time.
Keyword Tip: "ISO 13485 medical video" and "quality standards healthcare" are quality search keywords.
Infection control standards are critical guidelines designed to prevent the spread of harmful pathogens within healthcare environments. These standards, established by organizations like the CDC, WHO, and FDA, outline protocols for sanitation, sterilization, and the proper use of medical equipment. Adhering to these measures is essential in maintaining patient safety, reducing hospital-acquired infections (HAIs), and ensuring compliance with healthcare regulations.
Equipment used in sterile facilities, like operating rooms, cleanrooms, or labs, has to be designed particularly to be resistant to contamination and uphold stringent standards of hygiene. These settings require the highest standards of cleanliness to prevent bacterial and viral transfer, as well as other potentially toxic particles that may undermine patient safety or the integrity of sensitive procedures. Accordingly, video equipment should have seamless, easy-to-clean surfaces without any crevices in which contaminants can collect. Standards: ISO 14644 (cleanroom classification) and ASTM sterilization standards.
Key Requirements: Sealed designs, chemical resistance (e.g., to hydrogen peroxide), and autoclavable parts.
Healthcare Impact: Prevents infections like MRSA in the OR.
A cleanroom monitor wiped down mid-procedure remains sterile, covering a patient undergoing open-heart surgery.
Keyword Tip: "Sterile medical video equipment" and "cleanroom safety standards" resonate with OR teams.
Biocompatibility (ISO 10993) is essential for equipment used in patient care environments. This international standard ensures that materials in medical devices, including cleanroom monitors, are safe when in direct or indirect contact with human tissue. Compliance with ISO 10993 means the device has undergone rigorous testing for toxicity, irritation, and allergic response, making it suitable for sterile or surgical environments. For healthcare facilities, using biocompatible equipment supports infection control, patient safety, and adherence to global regulatory standards—critical in high-stakes clinical settings.
Purpose: Evades toxic reactions from materials.
Key Requirements: Testing for cytotoxicity, irritation, and sensitization.
Healthcare Impact: Makes sure a scope's tip doesn't hurt tissue.
A biocompatible camera prevents allergic reactions in a colonoscopy, increasing patient safety.
Keyword Tip: "ISO 10993 healthcare" and "biocompatible medical video" struck safety questions.
Data and cybersecurity standards within healthcare are necessary in guarding sensitive patient data and maintaining clinical system integrity.
These standards set best practices for data encryption, access controls, network security, and incident response procedures to protect against breaches, unauthorized access, and cyberattacks. Standards such as HIPAA, ISO/IEC 27001, and NIST offer formal guidelines for ensuring confidentiality, integrity, and availability of health information. By following these standards, healthcare organizations can establish a secure digital environment that not only complies with regulatory needs but also instills confidence among patients and stakeholders in an increasingly interconnected healthcare environment. 6. HIPAA Compliance
Patient information is frequently included in video feeds, so the Health Insurance Portability and Accountability Act (HIPAA) comes into play:
Purpose: Preserves privacy while recording or streaming.
Key Requirements: Encryption, access controls, and audit logs.
Healthcare Impact: Obtains live surgical webcasts to remote students.
A HIPAA-compliant monitor avoids a data breach, rescuing a hospital from a $2 million penalty.
Keyword Tip: "HIPAA medical video" and "secure healthcare imaging" attract privacy searches.
IEC 62304 sets the benchmark for software safety in medical devices. This international standard outlines the lifecycle requirements for the development and maintenance of medical software, ensuring it operates reliably and safely within clinical settings. For cleanroom monitors and imaging systems, compliance with IEC 62304 guarantees that embedded software is risk-managed, rigorously tested, and traceable throughout its updates. This level of control is crucial in healthcare, where even minor software errors can impact patient outcomes, making IEC 62304 a cornerstone of trustworthy, high-performance medical technology.
Purpose: Avoids crashes or errors during critical situations.
Key Requirements: Lifecycle management, testing, and error handling.
Healthcare Impact: Maintains a live feed stable throughout a trauma case.
An IEC 62304-validated system provides assurance that a monitor does not freeze during the procedure, thus preventing delays.
Keyword Tip: "Software safety medical video" and "IEC 62304 healthcare" extend reach.
The practical implications of integration and standardization in healthcare are extensive, extending directly to patient care, operational efficiency, and regulatory compliance.
Integrated systems allow health professionals to receive timely, accurate patient information departmentally, resulting in quicker diagnoses, better-informed treatment choices, and enhanced patient outcomes. Standardized processes and safety protocols provide consistency of care delivery, minimize the potential for errors, and facilitate compliance with regulatory mandates. Integrated workflows also eliminate administrative inefficiencies, minimize redundancy, and minimize operational expenses. On the cybersecurity front, standardized safeguards protect sensitive data from breaches, enhancing both patient trust and institutional resilience. Overall, the practical benefits of these systems translate into a safer, more efficient, and patient-centered healthcare environment. 8. Regulatory Compliance
FDA Approval: Required for U.S. market entry, aligning with IEC and ISO.
CE Marking: Mandatory for Europe, covering similar safety rules.
Audits: Facilities must prove compliance or face penalties.
A hospital that passes an FDA inspection attributes a clean slate to its compliant video equipment.
Keyword Tip: "FDA-approved medical video" and "healthcare compliance standards" inform regulatory searches.
Patient and staff safety are paramount in healthcare environments, and medical equipment must be designed to protect both. Monitors and imaging systems used in cleanrooms and surgical settings must prioritize electrical safety, radiation protection, and ergonomics. Features like radiation shielding, antimicrobial surfaces, and intuitive user interfaces reduce risks for both patients and medical personnel. Compliance with safety standards such as IEC 60601-1 and ISO 10993 ensures that equipment is safe to use, while minimizing the risk of contamination, exposure, or injury during operations. Safe, reliable equipment is critical to maintaining optimal patient care and protecting staff in high-risk environments.
Decreased Risks: No shocks, burns, or infections from equipment.
Reliability: Equipment functions under stress, such as a 12-hour surgery.
Training Ease: Safe equipment makes staff onboarding easier.
A nurse who uses a compliant monitor prevents a shock, concentrating on patient care instead.
Keyword Tip: "Safe medical video equipment" and "healthcare staff safety" emphasize outcomes.
Long-term reliability is crucial for medical equipment, especially in high-demand environments like cleanrooms and operating rooms. Monitors and imaging systems must be built to withstand frequent use, rigorous cleaning, and exposure to harsh conditions without compromising performance. Features such as durable construction, resistant coatings, and advanced cooling systems contribute to the longevity of the equipment. Choosing devices with proven reliability ensures that they will continue to perform accurately and consistently, reducing the need for costly repairs or replacements and supporting sustained patient care and clinical efficiency over time.
Durability: Resists cleaning and heavy wear.
Cost Savings: Breakdowns or replacements are fewer.
Future-Proofing: Compliant with changing standards through updates.
A $10,000 monitor that performs perfectly for 10 years trumps a $2,000 unit that crashes every year.
Keyword Tip: "Reliable medical video" and "long-lasting healthcare equipment" aim at durability.
When selecting medical video equipment, look for:
Certifications: IEC 60601-1, ISO 13485, FDA clearance.
Materials: Sterilizable (e.g., stainless steel), non-toxic (e.g., medical-grade plastic).
Software: Crash-proof, secure, and updatable.
Design: Ergonomic, sealed, and radiation-safe.
Support: Compliance documentation and service from vendor.
MedImaging's SafeView line, for example, checks all those boxes, combining safety with performance.
Keyword Tip: "Certified medical video equipment" and "safety features healthcare" power product searches.
Imagine an OR: A 4K endoscopy camera, IEC 60601-1 compliant, provides a consistent stream during a tumor removal—no flicker, no danger. In a cath lab, an ALARA-friendly fluoroscope reduces exposure time to radiation, shielding a cardiologist when placing a stent. Or when demonstrating a procedure in a training session, a HIPAA-compliant monitor broadcasts a live procedure to students while still maintaining patient privacy. These aren't fantasies—standards bring them to life each and every day.
Cost: Compliant equipment is more expensive ($5,000-$20,000 vs. $1,000 for non-medical). Solution: Account for ROI through safety and longevity.
Complexity: Standards overlap, confusing buyers. Solution: Collaborate with vendors such as MedImaging for advice.
Updates: Rules change, threatening obsolescence. Solution: Select modular, upgradable systems.
As technology improves, compliance gets easier and cheaper.
Research Standards: Match equipment to your use case (e.g., OR vs. lab).
Verify Certifications: Inspect FDA, CE, or ISO tags and papers.
Test in Context: Demo equipment in your own facility for safety and fit.
Train Staff: Educate correct use and upkeep to keep in compliance.
Partner with Experts: Rely on vendors for audits and upgrades.
Medical video equipment safety standards aren't just red tape—trust in healthcare's foundation. They safeguard against risk, maintain reliability, and protect data while improving care. Electrical safety to sterility to cybersecurity, these regulations enable clinicians to care, not troubleshoot. Ready to put safety first? Discover MedImaging compliant solutions and construct a safer, smarter healthcare ecosystem today.
Contact MedImaging today to schedule a compliance consultation, request a product demo, or receive a custom quote tailored to your facility's unique needs.
We are proud of the quality of service we provide for all orders and questions from customers. Our customer care team's combined experience and skills, together with our wide choice of products and quick order fulfilment, have helped us establish a solid reputation for providing excellent customer support & service.
Any Query Related Sales
09:00AM to 06:00PM
We welcome comments and feedback about how easy you find the Media k Solutions website to use, which can be communicated by clicking on the Contact Page. If you have questions about delivery, products, service or anything else for that matter, please contact us via live-chat, during office hours, email or telephone. Thanks
